Abstract
Purpose
To evaluate the safety and efficacy of intravitreal injection of bevacizumab advanced to vitrectomy for severe proliferative diabetic retinopathy.
Methods
Eight eyes of six patients (33–64 years old, all male patients) with severe proliferative diabetic retinopathy were investigated. An intravitreal injection of 1.25 mg bevacizumab was performed 3–30 days prior to planned vitrectomy.
Results
All cases showed minimum bleeding during surgical dissection of fibrovascular membrane. Two cases receiving bevacizumab 7 days before the surgery showed strong fibrosis and adhesion of fibrovascular membrane, resulted in some surgical complications. The cases having intravitreal bevacizumab for shorter time did not show extensive fibrosis.
Conclusions
The pretreatment of bevacizumab is likely effective in the vitrectomy for severe PDR. The appropriate timing of vitrectomy after bevacizumab injection should be further evaluated.
Similar content being viewed by others
Introduction
Proliferative diabetic retinopathy (PDR) is one of the most prevalent but severe ocular disorders, which is a major cause of adult blindness.1 To date, vitrectomy is a predominant treatment for PDR,1, 2 while the risk of complication is particularly concerned due to the bleeding from fibrovascular membrane (FVM).3 Bevacizumab (Avastin Genetech Inc., South San Francisco, California, USA) is a humanized antivascular endothelial growth factor (VEGF) antibody used for metastatic colorectal cancer.4 Recent reports have described the application of bevacizumab to treat ocular neovascular disorders including PDR.5, 6, 7, 8 Adjunctive use of intravitreal bevacizumab for severe PDR before vitrectomy has also been reported,5, 6 however, the preferable timing from the injection to surgery has not been determined. Here, we investigate eight eyes of six cases of severe PDR treated with intravitreal bevacizumab followed by vitrectomy and evaluate a suitable timing of pretreatment with bevacizumab.
Patients and methods
Patients with severe PDR were referred to our institute. The details of patients' data were shown in Table 1. They received routine ophthalmological examinations including fluorescein angiography (FA). We recruited only the patients with stage Vb or VI PDR by Scott's classification in this study.9 After extensive discussion of risks and benefits of intravitreal bevacizumab-assisted vitrectomy, each patient signed a comprehensive consent form. An intravitreal injection of 1.25 mg bevacizumab was followed by planned vitrectomy after 3–30 days.10
Results
All patients showed remarkable regression of FVM with visually absent vascular component in surgical procedures, which necessitated minimum intravitreal hemostasis and achieved complete resolution of tractional retinal detachment (TRD). An extensive fibrosis of FVM accompanying with very strong adhesion to the retina was found in two of three eyes, which received intravitreal bevacizumab seven days ahead of the surgery, whereas three eyes having shorter administration time of bevacizumab did not show firm adhesion of FVM to the retina. The followings are representative cases.
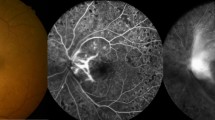
Patient 1: A 64-year-old man presented with visual disturbance in both eyes. Funduscopic examination revealed severe FVM on the optic disc and vascular arcades accompanied with segmental TRD in both eyes (Figure 1). Both eyes are followed by vitrectomy after a week of intravitreal injection of bevacizumab. Fluorescein angiography taken before the day of surgery showed a remarkable resolution of the leakage from FVM. During the surgical procedures, the vascular component of FVM looked retracted and little bleeding occurred, which necessitated no intraocular hemostasis throughout the surgery. However, extensive fibrosis was accompanied with strong adhesion of FVM to the retina and some retinal damages could not be avoided to accomplish the complete removal of FVM.
Fundus photographs and fluorescein angiography (FA) images of Case 1. (a) Left fundus at the initial visit shows fibrovascular membrane (FVM) surrounding the optic disc. (b) Same fundus one week after intravitreal injection of bevacizumab. The fibrosis of FVM is progressed (arrows). (c) Severe leakage of dye is found at neovascularization on the disc and elsewhere. (d) Same fundus shows remarkable attenuation of dye leakage at one-week postintravitreal bevacizumab.
Patient 4: A 43-year-old man, presented with visual disturbance in both eyes. Funduscopic examination revealed total retinal detachment with extensive fibrovascular proliferations on the retina (Figure 2). One week after the injection of bevacizumab in the left eye and three days after the injection in the right eye, vitrectomies were performed. Vascular components of FVM were remarkably retracted and minimum bleeding was found during the surgery in both eyes. Though an additional surgery was needed in the left eye for a recurrent TRD due to the traction of fibrotic tissues firmly adhered to the retina, both eyes achieved complete retinal reattachment.
Fundus photographs and fluorescein angiography (FA) images of Case 4. (a) Left fundus at the initial visit shows severe FVM accompanied with total retinal detachment. (b) Right fundus shows severe tractional retinal detachment (TRD) due to the contraction of ring- shaped FVM. (c) Left fundus after vitrectomy adjunct to intravitreal injection of bevacizumab achieves complete retinal reattachment. (d) Right fundus shows almost complete reattachment of the retina after the second surgery with silicone-oil tamponade for recurrent partial TRD due to severe fibrosis of FVM (arrows).
Discussion
We have presented six severe PDR cases, which were successfully treated with the combination of vitrectomy and advanced intravitreal bevacizumab. In the vitrectomy of severe PDR, we often encounter with sustained bleeding that obscures surgical field and prolongs surgical time, which results in the increase of the risk of accidental retinal damages. Avery et al5 have demonstrated the efficacy of bevacizumab for the treatment of PDR, which remarkably attenuates the activity of FVM at a week's postadministration. In fact, our cases showed minimum bleeding in surgical excisions of FVM even after three days of administration. It enabled the surgeons to keep good visibilities during the vitrectomies and reduced surgical risks. However, the excessive fibrosis of FVM may increase tissue traction as found in age-related macular degeneration cases.11 Actually, the generation of strong adhesion between FVM and retina was suspected in a few cases, which were administrated for bevacizumab a week before surgery. Since the cases received intravitreal bevacizumab for 3 to 5 days before surgery did not show extensive fibrosis of FVM, shorter administration period of bevacizumab may be preferable. Further studies are required to determine the appropriate timing of vitrectomy after intravitreal bevacizumab injection.
In conclusion, an appropriate use of intravitreal bevacizumab is likely effective as an adjunctive therapy ahead to vitrectomy for severe PDR.
References
Fong DS, Aiello LP, Ferris III FL, Klein R . Diabetic retinopathy. Diabetes Care 2004; 27 (10): 2540–2553.
Joussen AM, Joeres S . Benefits and limitations in vitreoretinal surgery for proliferative diabetic retinopathy and macular edema. Dev Ophthalmol 2007; 39: 69–87.
Helbig H, Kellner U, Bornfeld N, Foerster MH . Vitrectomy in diabetic retinopathy: outcome, risk factors, complications. Klin Monatsbl Augenheilkd (Written In German) 1998; 212 (5): 339–342.
Kramer I, Lipp HP . Bevacizumab, a humanized anti-angiogenic monoclonal antibody for the treatment of colorectal cancer. J Clin Pharm Ther 2007; 32 (1): 1–14.
Avery RL, Pearlman J, Pieramici DJ, Rabena MD, Castellarin AA, Nasir MA et al. Intravitreal bevacizumab (Avastin) in the treatment of proliferative diabetic retinopathy. Ophthalmology 2006; 113 (10): 1695.e1–1695.e15.
Chen E, Park CH . Use of intravitreal bevacizumab as a preoperative adjunct for tractional retinal detachment repair in severe proliferative diabetic retinopathy. Retina 2006; 26 (6): 699–700.
Mason III JO, Nixon PA, White MF . Intravitreal injection of bevacizumab (Avastin) as adjunctive treatment of proliferative diabetic retinopathy. Am J Ophthalmol 2006; 142 (4): 685–688.
Luke M, Januschowski K, Warga M, Beutel J, Leitritz M, Gelisken F et al. Intravitreal bevacizumab (Avastin) therapy for persistent diffuse diabetic macular edema. Retina 2006; 26 (9): 999–1005.
Scott GI . Ocular complications of diabetes mellitus. Br J Ophthalmol 1953; 37 (12): 705–715.
Beer PM, Wong SJ, Hammad AM, Falk NS, O'Malley MR, Khan S . Vitreous levels of unbound bevacizumab and unbound vascular endothelial growth factor in two patients. Retina 2006; 26 (8): 871–876.
Gibran SK, Sachdev A, Stappler T, Newsome R, Wong D, Hiscott P . Histological findings of a choroidal neovascular membrane removed at the time of macular translocation in a patient previously treated with intravitreal bevacizumab treatment (Avastin). Br J Ophthalmol 2007; 91 (5): 602–604.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ishikawa, K., Honda, S., Tsukahara, Y. et al. Preferable use of intravitreal bevacizumab as a pretreatment of vitrectomy for severe proliferative diabetic retinopathy. Eye 23, 108–111 (2009). https://doi.org/10.1038/sj.eye.6702983
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.eye.6702983
Keywords
This article is cited by
-
Pre-operative intravitreal bevacizumab for tractional retinal detachment secondary to proliferative diabetic retinopathy: the Alvaro Rodriguez lecture 2023
International Journal of Retina and Vitreous (2023)
-
Comparison of efficacy and safety of intravitreal ranibizumab and conbercept before vitrectomy in Chinese proliferative diabetic retinopathy patients: a prospective randomized controlled trial
Eye and Vision (2022)
-
Intravitreal bevacizumab (IVB) versus IVB in combination with pars plana vitrectomy for vitreous hemorrhage secondary to proliferative diabetic retinopathy: a randomized clinical trial
International Journal of Retina and Vitreous (2021)
-
Safety and efficacy of intravitreal conbercept injection after vitrectomy for the treatment of proliferative diabetic retinopathy
Eye (2019)
-
Frequency and Risk Factors for Neovascular Glaucoma After Vitrectomy in Eyes with Diabetic Retinopathy: An Observational Study
Diabetes Therapy (2019)