Abstract
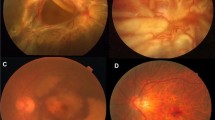
• Background: Scatter photocoagulation induces regression of retinal neovascularization, but the mechanism of its therapeutic effect is incompletely understood. To elucidate the mechanism of therapeutic effect of photocoagulation is the main focus of our research. We have already demonstrated basic fibroblast growth factor (bFGF) immunolocalization during retinal wound repair following laser photocoagulation. Transforming growth factor beta (TGF β) reportedly inhibits endothelial cell growth and bFGF-induced cell proliferation in vitro. In the present study, we evaluated the immunohistochemical localization of TGF-β1 and -β2 during wound repair in the rat retina following laser photocoagulation. • Methods: Krypton laser photocoagulation was performed on the eyes of pigmented rats. The eyes were then enucleated on day 1, 3, 7, 14, 28 or 56 following the photocoagulation and enrolled into the analysis of immunohistochemical localization of TGF-β1 and -β2. • Results: Immunoreactivity for TGF-β1 and -β2 was present in the ganglion cell layer and photoreceptor outer segments of the normal adult rat retina. The cytoplasm of RPE cells at the photocoagulated lesion showed intense TGF-β1 and -β2 immunoreactivity on day 3 after laser photocoagulation. Macrophages that migrated into the lesion lacked positive staining for TGF-β1 and -β2. TGF-β immunoreactivity in RPE cells continued to be upregulated for more than 1 month compared with that in normal RPE cells. Controls did not exhibit any positive staining. • Conclusion: An elevated expression of TGF-β immunoreactivity for a longer period of time than bFGF was observed in RPE cells at the photocoagulated lesion in vivo. In the late phase of retinal wound repair, TGF-β may inhibit cell proliferation induced by mitogens, introduce an end stage of cellular events, and induce extracellular matrix induction.
Similar content being viewed by others
References
Ammann AJ, Beck LS, Deguzman L, Hirabayashi SE, Lee WP, Mcfatridge L, Nguyen T, Xu Y (1990) Transforming growth factor-β: effect on soft tissue repair. Ann NY Acad Sci 593: 124–134
Anderson DH, Hageman GS, Guèrin CJ, Flanders KC (1991) Immunolocalization of transforming growth factor (TGF)-β1, TGF-β2, and TGF-β3 in mammalian retinas. Invest Ophthalmol Vis Sci [ARVO Suppl] 32: 754
Baird A, Durkin T (1986) Inhibition of endothelial cell proliferation by type β-transforming growth factor: interactions with acidic and basic fibroblast growth factors. Biochem Biophys Res Commun 138: 476–482
Baldin V, Rpman A-M, Bosc-Bierne I, Amalric F, Bouche G (1990) Translocation of bFGF to the nucleus is G1 phase cell cycle specific in bovine aortic endothelial cells. EMBO J 9: 1511–1517
Cromack DT, Sporn MB, Roberts AB, Merino MJ, Dart LL, Norton JA (1987) Transforming growth factor β levels in rat wound chambers. J Surg Res 42: 622–628
Fràter-Schröder M, Müller G, Birchmeier W, Böhlen P (1986) Transforming growth factor-beta inhibits endothelial cell proliferation. Biochem Biophys Res Commun 137: 295–302
Ignotz RA, Massague J (1986) Transforming growth factor-beta stimulates the expression of fibronectin and collagen and their incorporation into the extracellular matrix. J Biol Chem 261: 4337–4345
Lutty G, Ikeda K, Chandler C, McLeod DS (1991) Immunohistochemical localization of transforming growth factor-β in human photoreceptors. Curr Eye Res 10: 61–74
Lutty GA, Merges C, Threlkeld AB, Crone S, McLeod DS (1993) Heterogeneity in localization of isoforms of TGF-β in human retina, vitreous, and choroid. Invest Ophthalmol Vis Sci 34: 477–487
Lyons RM, Keski-Oja J, Moses HL (1988) Proteolytic activation of latent transforming growth factor-β from fibroblast-conditioned medium J Cell Biol 106: 1659–1665
Matsumoto M, Yoshimura N, Honda Y (1994) Increased production of transforming growth factor-β2 from cultured human retinal pigment epithelial cells by photocoagulation. Invest Ophthalmol Vis Sci 35: 4245–4252
Matsuoe TA, Pierce GF, Thomason A, Gramates P, Sporn MB, Deuel TF (1987) Accelerated healing of incisional wounds in rats induced by transforming growth factor-β. Science 237: 1333–1336
Miller DA, Pelton RW, Derynck R, Moses HL (1990) Transforming growth factor-β: a family of growth regulatory peptides. Ann NY Acad Sci 593: 208–217
Miyazono K, Hellman U, Wernstedt C, Heldin C-H (1988) Latent high molecular weight complex of transforming growth factor-β1. J Biol Chem 263: 6407–6415
Moses HL, Yang EY, Pietenpol JA (1990) TGF-β stimulation and inhibition of cell proliferation: new mechanistic insights. Cell 63: 245–247
Müller G, Behrens J, Nussbaumer U, Bohlen P, Birchmeier W (1987) Inhibitory action of transforming growth factor β on endothelial cells. Proc Natl Acad Sci USA 84: 5600–5604
Nakanishi Y, Kihara K, Mizuno K, Masamune Y, Yoshitake Y, Nishikawa K (1992) Direct effect of basic fibroblast growth factor on gene transcription in a cell-free system. Proc Natl Acad Sci USA 89: 5216–5220
Ogata N, Yamamoto C, Miyashiro M, Yamada H, Matsushima M, Uyama M (1997) Expression of transforming growth factor-β mRNA in experimental choroidal neovascularization. Curr Eye Res 16: 9–18
Pepper MS, Vassalli JD, Orci L, Montesano R (1993) Biphasic effect of transforming growth factor-β1 on in vitro angiogenesis. Exp Cell Res 204: 356–363
Pfeffer BA, Flanders KC, Guerin CJ, Danielpour D, Anderson DH (1994) Transforming growth factor beta 2 is the predominant isoform in the neural retina, retinal pigment epithelium-choroid and vitreous of the monkey eye. Exp Eye Res 59: 323–333
Robert F, Abe M, Mignatti P, Rifkin DB (1992) Basic fibroblast growth factorinduced activation of latent transforming growth factor β in endothelial cells: regulation of plasminogen activator activity. J Cell Biol 118: 901–909
Roberts AB, Sporn MB (1990) The transforming growth factor-βs. In: Sporn MB, Roberts AB (eds) Peptide growth factors and their receptors. (Handbook of experimental pharmacology, vol 95) Springer, Berlin Heidelberg New York, pp 419–472
Roberts AB, Sporn MB, Assoian RK, Smith JM, Roche NS, Wakefield LM, Heine UI, Liotta LA, Falanga V, Kehrl JH, Fauci AS (1986) Transforming growth factor β: rapid induction of fibrosis and angiogenesis in vivo and stimulation of collagen formation in vitro. Proc Natl Acad Sci USA 83: 4167–4171
Roberts AB, Flanders KC, Kondaiah P, Thompson NL, Obberghen-Schilling EV, Wakefield L, Rossi P, Crombrugghe B, Heine U, Sporn MB (1988) Transforming growth factor β: biochemistry and roles in embryogenesis, tissue repair and remodeling, and carcinogenesis. Recent Prog Horm Res 44: 155–197
Sporn MB, Roberts AB (1992) Transforming growth factor-β: recent progress and new challenges. J Cell Biol 119: 1017–1021
Tanihara H, Yoshida M, Matsumoto M, Yoshimura N (1993) Identification of transforming growth factor-β expressed in cultured human retinal pigment epithelial cells. Invest Ophthalmol Vis Sci 34: 413–419
Tripathi BJ, Park JK, Tripathi RC (1989) Extracellular release of tissue plasminogen activator is increased with the phagocytic activity of the retinal pigment epithelium. Invest Ophthalmol Vis Sci 30: 2470–2473
Wahl SM, Hunt DA, Wakefield LM, McCartney-Francis N, Wahl LM, Roberts AB, Sporn MB (1987) Transforming growth factor type β induces monocyte chemotaxis and growth factor production. Proc Natl Acad Sci USA 84: 5788–5792
Wakefield LM, Smith DM, Flanders KC, Sporn MB (1988) Latent transforming growth factor-β from human platelets. J Biol Chem 263: 7646–7654
Yamada T, Hara S, Tamai M: (1990) Immunohistochemical localization of cathepsin D in ocular tissue. Invest Ophthalmol Vis Sci 31: 1217–1223
Yamamoto C, Ogata N, Matsushima M, Takahashi K, Miyashiro M, Yamada H, Maeda H, Uyama M, Matsuzaki K (1996) Gene expressions of basic fibroblast growth factor and its receptor in healing of rat retina after laser photocoagulation. Jpn J Ophthalmol 40: 480–490
Yamamoto C, Ogata N, X Yi, Takahashi K, Miyashiro M, Yamada H, Uyama M, Matsuzaki K (1996) Immunolocalization of basic fibroblast growth factor during wound repair in rat retina after laser photocoagulation. Graefe’s Arch Clin Exp Ophthalmol 234: 695–702
Yoshimura N, Matsumoto M, Shimizu H, Mandai M, Hata Y, Ishibashi T (1995) Photocoagulated human retinal pigment epithelial cells produce an inhibitor of vascular endothelial cell proliferation. Invest Ophthalmol Vis Sci 36: 1686–1691
Author information
Authors and Affiliations
Additional information
This study was supported in part by The Science Research Promotion Fund of the Japan Private School Promotion Foundation (Dr. Uyama), and by a Grant-in-Aid for Developmental Scientific Research from the Ministry of Education, Science and Culture of the Japanese Government (Dr. Uyama)
Rights and permissions
About this article
Cite this article
Yamamoto, C., Ogata, N., Yi, X. et al. Immunolocalization of transforming growth factor β during wound repair in rat retina after laser photocoagulation. Graefe’s Arch Clin Exp Ophthalmol 236, 41–46 (1998). https://doi.org/10.1007/s004170050040
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s004170050040