Abstract
Background
Recent meta-analyses of randomized clinical trials (RCTs) demonstrated a higher risk of heart failure (HF) with the use of thiazolidinediones (TZDs). However, this effect may have been diluted by including active controls. Also, it is uncertain whether the risk of HF is similar with rosiglitazone and pioglitazone.
Objectives
This study quantified the risks of HF with the use of TZDs in patients with or at high risk of developing type 2 diabetes mellitus (DM), and evaluated differential effects by type of TZD. Secondarily, we evaluated risks of peripheral edema.
Methods
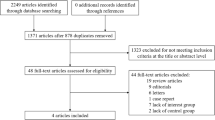
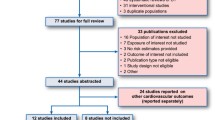
We performed a systematic review and meta-analysis of placebo-controlled RCTs evaluating the effect of rosiglitazone or pioglitazone on investigator-reported HF and edema. Articles published before 31 December 2009 were searched in MEDLINE, The Web of Science, and Scopus, and the data were extracted by three investigators. RCTs with ≥100 patients and ≥3 months of follow-up were included. We quantified the effect of TZDs as odds ratios (ORs) by using the Mantel-Haenzel and alternative models. We further evaluated the risk of serious/severe HF, and the effect of several trial characteristics on HF risk by subgroup analysis and meta-regression analysis.
Results
29 trials (n = 20254) were evaluated. TZDs were significantly associated with HF (TZD 360/6807 [5.3%] vs placebo 234/6328 [3.7%], OR 1.59; 95% CI 1.34, 1.89; p<0.00001). The risk of HF was higher with rosiglitazone than with pioglitazone (2.73 [95% CI 1.46, 5.10] vs 1.51 [1.26, 1.81]; p = 0.06). TZDs were associated with a similar risk of serious/severe HF (OR 1.47; 95% CI 1.16, 1.87; p = 0.002). Use of TZDs was also associated with edema (OR 2.04; 95% CI 1.85, 2.26; p<0.00001). HF and edema risks were consistent using Peto and random effects models. Risks of HF were significantly high for the subgroups of trials including patients with or at high risk for type 2 DM, and for the subgroup of trials with ≥12 months of follow-up. Meta-regression analysis showed that trials with lower overall baseline risk had higher HF risks.
Conclusion
In placebo-controlled trials of adult patients with or at high risk for type 2 DM, TZD therapy is significantly and consistently associated with a higher risk of HF. The risk of serious/severe HF is also increased with the use of TZDs. HF risks are similar to those of meta-analyses combining active- and placebo-controlled trials. The benefit/risk profile of TZDs should be considered when treating diabetic patients with or without prior HF.











Similar content being viewed by others
References
Selvin E, Bolen S, Yeh HC, et al. Cardiovascular outcomes in trials of oral diabetes medications: a systematic review. Arch Intern Med 2008; 168: 2070–80.
Nissen SE, Wolski K. Effect of rosiglitazone on the risk of myocardial infarction and death from cardiovascular causes. N Engl J Med 2007; 356: 2457–71.
Lincoff AM, Wolski K, Nicholls SJ, et al. Pioglitazone and risk of cardiovascular events in patients with type 2 diabetes mellitus: a meta-analysis of randomized trials. JAMA 2007; 298: 1180–8.
Singh S, Loke TK, Furberg CD. Long-term risk of cardiovascular events with rosiglitazone: a meta-analysis. JAMA 2007; 298: 1189–95.
Lago RM, Singh PP, Nesto RW. Congestive heart failure and cardiovascular death in patients with pre-diabetes and type 2 diabetes given thiazolidinediones: a meta-analysis of randomized clinical trials. Lancet 2007; 370: 1129–36.
Berlie HD, Kalus JS, Jaber LA. Thiazolidinediones and the risk of edema: A meta-analysis. Diabetes Res Clin Pract 2007; 76: 279–89.
Karalliedde J, Buckingham RE. Thiazolidinediones and their fluid-related adverse effects: facts, fiction and putative management strategies. Drug Saf 2007; 30: 741–53.
Buckingham RE, Hanna A. Thiazolidinedione insulin sensitizers and the heart: a tale of two organs? Diabetes Obes Metab 2008; 10: 312–28.
St John Sutton M, Rendell M, Dandona P, et al. A comparison of the effects of rosiglitazone and glyburide on cardiovascular function and glycemic control in patients with type 2 diabetes. Diabetes Care 2002; 25: 2058–64.
Kannel WB, Hjortland M, Castelli WP. Role of diabetes in congestive heart failure: the Framingham Study. Am J Cardiol 1974; 34: 29–34.
Tang WH. Do thiazolidinediones cause heart failure? A critical review. Cleve Clinic J Med 2006; 73: 390–6.
Mannucci E, Monami M, Di Bari M, et al. Cardiac safety profile of rosiglitazone: a comprehensive meta-analysis of randomized clinical trials. Int J Cardiol 2009. doi: 10.1016/j.ijcard.2009.01.064.
Johnson JA, Majumdar SR, Simpson SH, et al. Decreased mortality associated with the use of metformin compared with sulfonylurea monotherapy in type 2 diabetes. Diabetes Care 2002; 25: 2244–8.
Koro CE, Bowlin SJ, Weiss SR. Antidiabetic therapy and the risk of heart failure in type 2 diabetic patients: an independent effect or confounding by indication. Pharmacoepidemiol Drug Saf 2005; 14: 697–703.
Hernandez AV, Walker E, Ioannidis JP, et al. Challenges in meta-analysis of randomized clinical trials for rare harmful cardiovascular events: the case of rosiglitazone. Am Heart J 2008; 156: 23–30.
Sweeting MJ, Sutton AJ, Lambert PC. What to add to nothing? Use and avoidance of continuity corrections in meta-analysis of sparse data. Stat Med 2004; 23: 1351–75.
Bradburn MJ, Deeks JJ, Berlin JA, et al. Much ado about nothing: a comparison of the performance of meta-analytical methods with rare events. Stat Med 2007; 26: 53–77.
Hernandez AV, Westerhout CM, Steyerberg EW, et al. Effects of platelet glycoprotein IIb/IIIa receptor blockers in non-ST segment elevation acute coronary syndromes: benefit and harm in different age subgroups. Heart 2007; 93: 450–5.
Jadad AR, Moore RA, Carroll D, et al. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials 1996; 17: 1–12.
Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med 2009; 151: 264–9.
Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med 2002; 21: 1539–58.
Egger M, Altman DG, Smith GD. Systematic reviews in health-care: metaanalysis in context. 2nd ed. London: BMJ Books, 2001.
Yusuf S, Peto R, Lewis J, et al. Beta blockade during and after myocardial infarction: an overview of the randomized trials. Prog Cardiovasc Dis 1985; 27: 335–71.
DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials 1986; 7: 177–88.
van Houwelingen HC, Arends LR, Stijnen T. Advanced methods in metaanalysis: multivariate approach and meta-regression. Stat Med 2002; 21: 589–624.
Agrawal A, Sautter MC, Jones NP. Effects of rosiglitazone maleate when added to a sulfonylurea regimen in patients with type 2 diabetes mellitus and mild to moderate renal impairment: a post hoc analysis. Clin Ther 2003; 25: 2754–64.
Albertini J-P, McMorn SO, Chen H, et al. Effect of rosiglitazone on factors related to endothelial dysfunction in patients with type 2 diabetes mellitus. Atherosclerosis 2007; 195: e159–66.
Barnett AH, Grant PJ, Hitman GA, et al. Rosiglitazone in type 2 diabetes: an evaluation in British Indo-Asian patients. Diabet Med 2003; 20: 387–93.
Bhatt DL, Chew DP, Grines C, et al. Peroxisome proliferator-activated receptor γ agonists for the prevention of adverse events following percutaneous coronary revascularization: results of the PPAR Study. Am Heart J 2007; 154: 137–43.
Cao Z, Zhou YJ, Zhao YX, et al. Rosiglitazone could improve clinical outcomes after coronary stent implantation in non-diabetic patients with metabolic syndrome. Chin Med J 2006; 119: 1171–5.
Dailey GE, Noor MA, Park J-S, et al. Glycemic control with glyburide/ metformin tablets in combination with rosiglitazone in patients with type 2 diabetes: a randomized double-blind trial. Am J Med 2004; 116: 223–9.
Dargie HJ, Hildebrandt PR, Riegger GA, et al. A randomized, placebo-controlled trial assessing the effects of rosiglitazone on echocardiographic function and cardiac status in type 2 diabetic patients with New York Heart Association functional class I or II heart failure. J Am Coll Cardiol 2007; 49: 1696–704.
Davidson JA, McMorn SO, Waterhouse BR, et al. A 24 week, multicenter, randomized, double-blind, placebo-controlled, parallel-group study of the efficacy and tolerability of combination therapy with rosiglitazone and sulfonylurea in African American and Hispanic American patients with type 2 diabetes inadequately controlled with sulfonylurea monotherapy. Clin Ther 2007; 29: 1900–14.
Fonseca V, Rosenstock J, Patwardhan R, et al. Effect of metformin and rosiglitazone combination therapy in patients with type 2 diabetes mellitus: a randomized controlled trial. JAMA 2000; 283: 1695–702.
Gerstein HC, Yusuf S, Bosch J, et al. Effect of rosiglitazone on the frequency of diabetes in patients with impaired glucose tolerance or impaired fasting glucose: a randomised controlled trial. Lancet 2006; 368: 1096–105.
Gómez-Perez FJ, Fanghänel-Salmón G, Barbosa JA, et al. Efficacy and safety of rosiglitazone plus metformin in Mexicans with type 2 diabetes. Diabetes Metab Res Rev 2002; 18: 127–34.
Hedblad B, Zambanini A, Nilsson P, et al. Rosiglitazone and carotid IMT progression rate in a mixed cohort of patients with type 2 diabetes and the insulin resistance syndrome: main results from the Rosiglitazone Atherosclerosis Study. J Intern Med 2007; 261: 293–305.
Hollander P, Yu D, Chou HS. Low-dose rosiglitazone in patients with insulinrequiring type 2 diabetes. Arch Intern Med 2007; 167: 1284–90.
Lebovitz HE, Dole JF, Patwhardhan R, et al., for the Rosiglitazone Clinical Trials Study Group. Rosiglitazone monotherapy is effective in patients with type 2 diabetes. J Clin Endocrinol Metab 2001; 86: 280–8.
Phillips LS, Grunberger G, Miller E, et al. Once- and twice-daily dosing with rosiglitazone improves glycemia control in patients with type 2 diabetes. Diabetes Care 2001; 24: 308–15.
Raskin P, Rendell M, Riddle MC, et al. A randomized trial of rosiglitazone therapy in patients with inadequately controlled insulin-treated type 2 diabetes. Diabetes Care 2001; 24: 1226–32.
Rosenstock J, Goldstein BJ, Vinik AI, et al. Effect of early addition of rosiglitazone to sulphonylurea therapy in older type 2 diabetes patients (>60 years): the Rosglitazone Early vs SULphonylurea Titration (RESULT) study. Diabetes Obes Metab 2006; 8: 49–57.
Zhu XX, Pan CY, Li GW, et al. Addition of rosiglitazone to existing sulfonylurea treatment in Chinese patients with type 2 diabetes and exposure to hepatitis B or C. Diabetes Technol Ther 2003; 5: 33–42.
Aronoff S, Rosenblatt S, Braithwaite S, et al. Pioglitazone hydrochloride monotherapy improves glycemic control in the treatment of patients with type 2 diabetes: a 6-month randomized placebo-controlled dose-response study. Diabetes Care 2000; 23: 1605–11.
Berhanu P, Perez A, Yu S. Effect of pioglitazone in combination with insulin therapy on glycaemic control, insulin dose requirement and lipid profile in patients with type 2 diabetes previously poorly controlled with combination therapy. Diabetes Obes Metab 2007; 9: 512–20.
Einhorn D, Rendell M, Rosenzweig J, et al. Pioglitazone hydrochloride in combination with metformin in the treatment of type 2 diabetes mellitus: a randomized, placebo-controlled study. The Pioglitazone 027 Study Group. Clin Ther 2000; 22: 1395–409.
Erdmann E, Charbonnel B, Wilcox RG, et al. Pioglitazone use and heart failure in patients with type 2 diabetes and pre-existing cardiovascular disease: data from the PRO active study (PROactive 08). Diabetes Care 2007; 30: 2773–8.
Goldstein BJ, Rosenstock J, Anzalone D, et al. Effect of tesaglitazar, a dual PPARα/γ agonist, on glucose and lipid abnormalities in patients with type 2 diabetes: a 12-week dose-ranging trial. Curr Med Res Opin 2006; 22: 2575–90.
Herz M, Johns D, Reviriego J, et al. A randomized, double-blind, placebocontrolled, clinical trial of the effects of pioglitazone on glycemic control and dyslipidemia in oral antihyperglycemic medication-naïve patients with type 2 diabetes mellitus. Clin Ther 2003; 25: 1074–95.
Kipnes MS, Krosnick A, Rendell MS, et al. Pioglitazone hydrochloride in combination with sulfonylurea therapy improves glycemic control in patients with type 2 diabetes mellitus: a randomized, placebo-controlled study. Am J Med 2001; 111: 10–7.
Mattoo V, Eckland D, Widel M, et al. Metabolic effect of pioglitazone in combination with insulin in patients with type 2 diabetes mellitus whose disease is not adequately controlled with insulin therapy: results of a six-month, randomized, double-blind, prospective, multicenter, parallel-group study. Clin Ther 2005; 27: 554–67.
Rosenblatt S, Miskin B, Glazer NB, et al., Pioglitazone 026 Study Group. The impact of pioglitazone on glycemic control and atherogenic dyslipidemia in patients with type 2 diabetes mellitus. Coron Artery Dis 2001; 12: 413–23.
Rosenstock J, Einhorn D, Hershon K, et al., and the Pioglitazone 014 Study Group. Efficacy and safety of pioglitazone in type 2 diabetes: a randomised, placebo-controlled study in patients receiving stable insulin therapy. Int J Clin Pract 2002; 56: 251–7.
Scherbaum WA, Göke B, and the German Pioglitazone Study Group. Metabolic efficacy and safety of once-daily pioglitazone monotherapy in patients with type 2 diabetes: a double-blind, placebo-controlled study. Horm Metab Res 2002; 34: 589–95.
Temple R, Ellenberg SS. Placebo-controlled trials and active-control trials in the evaluation of new treatments. Ann Intern Med 2000; 133: 455–63.
Singh S, Loke YK, Furberg CD. Thiazolidinediones and heart failure: a teleoanalysis. Diabetes Care 2007; 30: 2148–53.
Mannucci E, Monami M, Lamanna C, et al. Pioglitazone and cardiovascular risk: a comprehensive meta-analysis of randomized clinical trials. Diabetes Obes Metab 2008; 10: 1221–38.
Richter B, Bandeira-Echtler E, Bergerhoff K, et al. Rosiglitazone for type 2 diabetes mellitus. Cochrane Database of Systematic Reviews 2007; Issue 3: CD006063. doi: 10.1002/14651858.CD006063.pub2.
Richter B, Bandeira-Echtler E, Bergerhoff K, et al. Pioglitazone for type 2 diabetes mellitus. Cochrane Database of Systematic Reviews 2007; Issue 4: CD006060. doi: 10.1002/14651858.CD006060.pub2.
Winkelmayer WC, Setoguchi S, Levin R, et al. Comparison of cardiovascular outcomes in elderly patients with diabetes who initiated rosiglitazone vs pioglitazone therapy. Arch Intern Med 2008; 168: 2368–75.
Juurlink DN, Gomes T, Lipscombe LL, et al. Adverse cardiovascular events during treatment with pioglitazone and rosiglitazone: population based cohort study. BMJ 2009; 339: b2942.
Graham DJ, Ouellet-Hellstrom R, MaCurdy TE, et al. Risk of acute myocardial infarction, stroke, heart failure, and death in elderly Medicare patients treated with rosiglitazone or pioglitazone. JAMA 2010; 304: 411–8.
Guan Y, Hao C, Cha DR, et al. Thiazolidinediones expand body fluid volume through PPARgamma stimulation of ENaC-mediated renal salt absorption. Nat Med 2005; 11: 861–6.
Wertz DA, Chang CL, Sarawate CA, et al. Risk of cardiovascular events and all-cause mortality in patients treated with thiazolidinediones in a managedcare population. Circ Cardiovasc Qual Outcomes 2010; 3: 538–45.
Rutter MK, Parise H, Benjamin EJ, et al. Impact of glucose intolerance and insulin resistance on cardiac structure and function: sex-related differences in the Framingham Heart Study. Circulation 2003; 107: 448–54.
Hutton JL. Number needed to treat and number needed to harm are not the best way to report and assess the results of randomised clinical trials. Br J Haematol 2009; 146: 27–30.
McAlister FA. The “number needed to treat” turns 20 — and continues to be used and misused. CMAJ 2008; 179: 549–53.
Massel D. The number need to harm: is it too optimistic? Can J Cardiol 2003; 19: 1490–2.
Loke YK, Singh S, Furberg CD. Long-term use of thiazolidinediones and fractures in type 2 diabetes: a meta-analysis. CMAJ 2009; 180: 32–9.
Nathan DM, Buse JB, Davidson MB, et al. Medical management of hyperglycemia in type 2 diabetes: a consensus algorithm for the initiation and adjustment of therapy. A consensus statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care 2009; 32: 193–203.
Home PD, Pocock SJ, Beck-Nielsen H, et al. Rosiglitazone evaluated for cardiovascular outcomes in oral agent combination therapy for type 2 diabetes (RECORD): amulticentre, randomised, open-label trial. Lancet 2009; 373: 2125–35.
US FDA Drug Safety and Availability. Postmarket drug safety information for patients and providers [online]. Available from URL: http: //www.fda.gov/ Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/ ucm226976.htm [Accessed 2010 Dec 8].
European Medicines Agency press release. European Medicines Agency recommends suspension of Avandia, Avandamet and Avaglim [online]. Available from URL: http: //www.ema.europa.eu/docs/en_GB/document_library/ Press_release/2010/09/WC500096996.pdf [Accessed 2010 Dec 8].
Acknowledgments
No external funding was used to support this work. The authors are solely responsible for the design and conduct of this study, all study analyses, the drafting and editing of the manuscript, and its final contents. The authors have no relevant conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hernandez, A.V., Usmani, A., Rajamanickam, A. et al. Thiazolidinediones and Risk of Heart Failure in Patients with or at High Risk of Type 2 Diabetes Mellitus. Am J Cardiovasc Drugs 11, 115–128 (2011). https://doi.org/10.2165/11587580-000000000-00000
Published:
Issue Date:
DOI: https://doi.org/10.2165/11587580-000000000-00000