Abstract
Aims
Frequent glucose testing is required for optimal management of type 1 diabetes (T1D). Limited data are available regarding real-world experience of the novel technology for monitoring by continuous interstitial fluid glucose (IFG), using flash glucose-sensing technology (FSL-CGM). We aimed to assess the effect of FSL-CGM in a real-life clinical setting on glycemic control parameters, compliance, and adverse events among pediatric and young adult T1D patients.
Methods
This observational multi-center study assessed FSL-CGM use (6–12 months) in T1D patients (mean ± SD age 13.4 ± 4.9 years) who purchased the device out-of-pocket. Outcome measures included HbA1c, mean IFG levels, CGM metrics [time in hypoglycemia (< 54 mg/dL; < 3 mmol/L), in target range (70–180 mg/dL; 3.9–10 mmol/L), and in hyperglycemia > 240 mg/dL; > 13.3 mmol/L)], frequency of self-monitoring of blood glucose, acute complications, skin reactions, and reasons for initiation/discontinuation.
Results
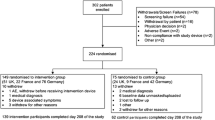
Among patients with regular use of the FSL-CGM (n = 59), mean HbA1c decreased from 8.86 ± 0.23 to 8.05 ± 0.2% (73.3–64.5 mmol/mol) in 3 months (p = 0.0001) and plateaued thereafter. A clinically significant reduction in HbA1c (defined as a decrease of ≥ 0.5%) was associated with shorter diabetes duration. Of 71 patients who initiated use of the FSL-CGM, 12 (16.9%) discontinued during the study period. No statistically significant changes were found after FSL-CGM use, in mean and standard deviation IFG levels, and in time of glucose levels in target, hypoglycemia, and hyperglycemia ranges. One patient with hypoglycemia unawareness was found dead-in-bed while using FSL-CGM.
Conclusions
Real-life observational data in a self-selected young T1D population demonstrated a significant and sustained reduction in HbA1c with FSL-CGM in one-third of the participants. Surveillance of glucose monitoring should be individualized, especially for patients with hypoglycemia unawareness.


Similar content being viewed by others
Change history
12 September 2018
Unfortunately, the co-author name was misspelled as “Avivit Brenner” instead of “Avivit Brener” in the original publication and the correct author name is updated here
References
Ziegler R, Heidtmann B, Hilgard D et al (2011) Frequency of SMBG correlates with HbA1c and acute complications in children and adolescents with type 1 diabetes. Pediatr Diabetes 12:11–17. https://doi.org/10.1111/j.1399-5448.2010.00650.x
Miller KM, Beck RW, Bergenstal RM et al (2013) Evidence of a strong association between frequency of self-monitoring of blood glucose and hemoglobin A1c levels in T1D exchange clinic registry participants. Diabetes Care 36:2009–2014. https://doi.org/10.2337/dc12-1770
Bergenstal RM, Tamborlane WV, Ahmann A et al (2010) Effectiveness of sensor-augmented insulin-pump therapy in type 1 diabetes. N Engl J Med 363:311–320. https://doi.org/10.1056/NEJMoa1002853
Juvenile Diabetes Research Foundation Continuous Glucose Monitoring Study Group, Tamborlane WV, Beck RW et al (2008) Continuous glucose monitoring and intensive treatment of type 1 diabetes. N Engl J Med 359:1464–1476. https://doi.org/10.1056/NEJMoa0805017
Charleer S, Mathieu C, Nobels F et al (2018) Effect of continuous glucose monitoring on glycemic control, acute admissions and quality of life: a real-world study. J Clin Endocrinol Metab. https://doi.org/10.1210/jc.2017-02498
Hermanides J, Nørgaard K, Bruttomesso D et al (2011) Sensor-augmented pump therapy lowers HbA1c in suboptimally controlled Type 1 diabetes; a randomized controlled trial. Diabet Med 28:1158–1167. https://doi.org/10.1111/j.1464-5491.2011.03256.x
Weiss R, Garg SK, Bode BW et al (2015) Hypoglycemia reduction and changes in hemoglobin A1c in the ASPIRE in-home study. Diabetes Technol Ther 17:542–547. https://doi.org/10.1089/dia.2014.0306
Battelino T, Nimri R, Dovc K et al (2017) Prevention of hypoglycemia with predictive low glucose insulin suspension in children with type 1 diabetes: a randomized controlled trial. Diabetes Care 40:764–770. https://doi.org/10.2337/dc16-2584
Wong JC, Foster NC, Maahs DM et al (2014) Real-time continuous glucose monitoring among participants in the T1D Exchange clinic registry. Diabetes Care 37:2702–2709. https://doi.org/10.2337/dc14-0303
Rachmiel M, Landau Z, Boaz M et al (2015) The use of continuous glucose monitoring systems in a pediatric population with type 1 diabetes mellitus in real-life settings: the AWeSoMe Study Group experience. Acta Diabetol 52:323–329. https://doi.org/10.1007/s00592-014-0643-6
Juvenile Diabetes Research Foundation Continuous Glucose Monitoring Study Group (2010) Effectiveness of continuous glucose monitoring in a clinical care environment: evidence from the Juvenile Diabetes Research Foundation continuous glucose monitoring (JDRF-CGM) trial. Diabetes Care 33:17–22. https://doi.org/10.2337/dc09-1502
Edge J, Acerini C, Campbell F et al (2017) An alternative sensor-based method for glucose monitoring in children and young people with diabetes. Arch Dis Child 102:543–549. https://doi.org/10.1136/archdischild-2016-311530
Bonora B, Maran A, Ciciliot S et al (2016) Head-to-head comparison between flash and continuous glucose monitoring systems in outpatients with type 1 diabetes. J Endocrinol Investig 39:1391–1399. https://doi.org/10.1007/s40618-016-0495-8
Al Hayek AA, Robert AA, Al Dawish MA (2017) Evaluation of FreeStyle libre flash glucose monitoring system on glycemic control, health-related quality of life, and fear of hypoglycemia in patients with type 1 diabetes. Clin Med Insights Endocrinol Diabetes 10:1179551417746957. https://doi.org/10.1177/1179551417746957
Bolinder J, Antuna R, Geelhoed-Duijvestijn P et al (2016) Novel glucose-sensing technology and hypoglycaemia in type 1 diabetes: a multicentre, non-masked, randomised controlled trial. Lancet 388:2254–2263. https://doi.org/10.1016/S0140-6736(16)31535-5
Dunn TC, Xu Y, Hayter G, Ajjan RA (2018) Real-world flash glucose monitoring patterns and associations between self-monitoring frequency and glycaemic measures: a European analysis of over 60 million glucose tests. Diabetes Res Clin Pract 137:37–46. https://doi.org/10.1016/j.diabres.2017.12.015
Szadkowska A, Gawrecki A, Michalak A et al (2017) Flash glucose measurements in children with type 1 diabetes in real-life settings: to trust or not to trust? Diabetes Technol Ther. https://doi.org/10.1089/dia.2017.0287
Massa GG, Gys I, Op ‘t Eyndt A et al (2018) Evaluation of the FreeStyle® libre flash glucose monitoring system in children and adolescents with type 1 diabetes. Horm Res Paediatr 89:189–199. https://doi.org/10.1159/000487361
Dover AR, Stimson RH, Zammitt NN, Gibb FW (2017) Flash glucose monitoring improves outcomes in a type 1 diabetes clinic. J Diabetes Sci Technol 11:442–443. https://doi.org/10.1177/1932296816661560
McKnight JA, Gibb FW (2017) Flash glucose monitoring is associated with improved glycaemic control but use is largely limited to more affluent people in a UK diabetes centre. Diabet Med 34:732–732. https://doi.org/10.1111/dme.13315
Ish-Shalom M, Wainstein J, Raz I, Mosenzon O (2016) Improvement in glucose control in difficult-to-control patients with diabetes using a novel flash glucose monitoring device. J Diabetes Sci Technol 10:1412–1413. https://doi.org/10.1177/1932296816653412
Haak T, Hanaire H, Ajjan R et al (2017) Use of flash glucose-sensing technology for 12 months as a replacement for blood glucose monitoring in insulin-treated type 2 diabetes. Diab Ther 8:573–586. https://doi.org/10.1007/s13300-017-0255-6
Duca LM, Wang B, Rewers M, Rewers A (2017) Diabetic ketoacidosis at diagnosis of type 1 diabetes predicts poor long-term glycemic control. Diabetes Care 40:1249–1255. https://doi.org/10.2337/dc17-0558
Reddy M, Jugnee N, El Laboudi A et al (2018) A randomized controlled pilot study of continuous glucose monitoring and flash glucose monitoring in people with type 1 diabetes and impaired awareness of hypoglycaemia. Diabet Med 35:483–490. https://doi.org/10.1111/dme.13561
Bolinder J, Antuna R, Geelhoed-Duijvestijn P et al (2017) Cutaneous adverse events related to FreeStyle libre device—authors’ reply. Lancet 389:1396–1397. https://doi.org/10.1016/S0140-6736(17)30893-0
Herman A, Aerts O, Baeck M et al (2017) Allergic contact dermatitis caused by isobornyl acrylate in Freestyle® Libre, a newly introduced glucose sensor. Contact Dermat 77:367–373. https://doi.org/10.1111/cod.12866
Englert K, Ruedy K, Coffey J et al (2014) Skin and adhesive issues with continuous glucose monitors: a sticky situation. J Diabetes Sci Technol 8:745–751. https://doi.org/10.1177/1932296814529893
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflicts of interest.
Human and animal rights
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the 1975 Declaration of Helsinki, as revised in 2008.
Informed consent
Each center obtained local ethics committee approval before enrollment. Because there was no identification of the patients for whom data were retrieved, informed consent by the patients was waived.
Additional information
Managed by Massimo Federici.
The original version of this article was revised: The co-author name was misspelled as “Avivit Brenner” instead of “Avivit Brener” in the original publication.
Rights and permissions
About this article
Cite this article
Landau, Z., Abiri, S., Gruber, N. et al. Use of flash glucose-sensing technology (FreeStyle Libre) in youth with type 1 diabetes: AWeSoMe study group real-life observational experience. Acta Diabetol 55, 1303–1310 (2018). https://doi.org/10.1007/s00592-018-1218-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-018-1218-8