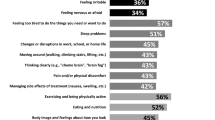
Abstract
Purpose
The aim of this study was to estimate the minimal clinical important difference (MCID) of the World Health Organization Quality of Life assessment instrument (WHOQOL-100) for women with early-stage breast cancer. If the MCID is known, then the instrument is also useful for individual patients and thus useful in clinical practice. In this study, the MCID is the smallest change in quality of life (QOL) scores considered clinically relevant for patients.
Methods
Data were derived from a prospective study in which 223 women with early-stage breast cancer and 383 women with benign breast problems completed the WHOQOL-100. MCID values were determined by two distribution-based methods (0.5 standard deviation (SD) and 1 standard error of measurement (SEM)) and an anchor-based method.
Results
For 0.5 SD, MCID estimates ranged from 0.64 to 0.94, and for 1 SEM, MCID estimates ranged from 0.69 to 1.18. The anchor-based approach resulted in MCID estimates ranging from −1.56 to −0.71 for decline and from 0.51 to 1.27 for improvement.
Conclusion
A change in score of 1 on the WHOQOL-100 is proposed as the MCID for women with early-stage breast cancer.
Similar content being viewed by others
References
WHOQOL group (1995) The World Health Organization Quality of Life assessment [WHOQOL]: position paper from the World Health Organization. Soc Sci Med 41:1403–1409
WHOQOL Group (1998) The World Health Organization Quality of Life Assessment [WHOQOL]: development and general psychometric properties. Soc Sci Med 46:1569–1585
Tazaki M, Nakane Y, Endo T, Kakikawa F, Kano K, Kawano H et al (1998) Results of a qualitative and field study using the WHOQOL instrument for cancer patients. Jpn J Clin Oncol 28:134–141
Den Oudsten BL, Van Heck GL, Van der Steeg AF, Roukema JA, De Vries J (2009) The WHOQOL-100 has good psychometric properties in breast cancer patients. J Clin Epidemiol 62(2):195–205
O'Carroll RE, Smith K, Couston M, Cossar JA, Hayes PC (2000) A comparison of the WHOQOL-100 and the WHOQOL-BREF in detecting change in quality of life following liver transplantation. Qual Life Res 9:121–124
O'Carroll RE, Couston M, Cossar J, Masterton G, Hayes PC (2003) Psychological outcome and quality of life following liver transplantation: a prospective, national, single-center study. Liver Transpl 9:712–720
Skevington SM, Carse MS, Williams AC (2001) Validation of the WHOQOL-100: pain management improves quality of life for chronic pain patients. Clin J Pain 17:264–275
Masthoff ED, Trompenaars FJ, Van Heck GL, Hodiamont PP, De Vries J (2005) Validation of the WHO Quality of Life assessment instrument [WHOQOL-100] in a population of Dutch adult psychiatric outpatients. Eur Psychiatry 20:465–473
Revicki D, Hays RD, Cella D, Sloan J (2008) Recommended methods for determining responsiveness and minimally important differences for patient-reported outcomes. J Clin Epidemiol 61(2):102–109
Sloan JA, Cella D, Frost M, Guyatt GH, Sprangers M, Symonds T (2002) Assessing clinical significance in measuring oncology patient quality of life: introduction to the symposium, content overview, and definition of terms. Mayo Clin Proc 77(4):367–370
Revicki DA, Cella D, Hays RD, Sloan JA, Lenderking WR, Aaronson NK (2006) Responsiveness and minimal important differences for patient reported outcomes. Health Qual Life Outcomes 4:70
De Vries J, Van Heck GL (1997) The World Health Organization Quality of Life assessment instrument [WHOQOL-100]: validation study with the Dutch version. Eur J Psychol Assess 13:164–178
Guyatt G, Schunemann H (2007) How can quality of life researchers make their work more useful to health workers and their patients? Qual Life Res 16(7):1097–1105
Norman GR, Sloan JA, Wyrwich KW (2003) Interpretation of changes in health-related quality of life: the remarkable universality of half a standard deviation. Med Care 41(5):582–592
Norman GR, Sloan JA, Wyrwich KW (2004) The truly remarkable universality of half a standard deviation: confirmation through another look. Expert Rev Pharmacoecon Outcomes Res 4(5):581–585
Crosby RD, Kolotkin RL, Williams GR (2003) Defining clinically meaningful change in health-related quality of life. J Clin Epidemiol 56(5):395–407
Bonomi AE, Patrick DL, Bushnell DM, Martin M (2000) Validation of the United States' version of the World Health Organization Quality of Life [WHOQOL] instrument. J Clin Epidemiol 53(1):1–12
O'Carroll RE, Cossar JA, Couston MC, Hayes PC (2000) Sensitivity to change following liver transplantation: a comparison of three instruments that measure quality of life. J Health Psychol 5:69–74
van der Steeg AF, De Vries J, Roukema JA (2010) Anxious personality and breast cancer: possible negative impact on quality of life after breast-conserving therapy. World J Surg 34(7):1453–1460
Van der Steeg AF, De Vries J, Van der Ent FW, Roukema JA (2007) Personality predicts quality of life six months after the diagnosis and treatment of breast disease. Ann Surg Oncol 14:678–685
Power M, Harper A, Bullinger M (1999) The World Health Organization WHOQOL-100: tests of the universality of Quality of Life in 15 different cultural groups worldwide. Health Psychol 18:495–505
WHOQOL Group (1998) Development of the World Health Organization WHOQOL-BREF quality of life assessment. The WHOQOL Group. Psychol Med 28:551–558
Spielberger CD, Gorsuch RL, Lushene RE (1970) STAI manual for the State-Trait Anxiety Inventory. Consulting Psychologists, Palo Alto
Van der Ploeg HM, Defares PB, Spielberger CD (1980) ZBV: a Dutch-language adaptation of the Spielberger State-Trait Anxiety Inventory. Swets & Zeitlinger, Lisse
Copay AG, Subach BR, Glassman SD, Polly DW Jr, Schuler TC (2007) Understanding the minimum clinically important difference: a review of concepts and methods. Spine J 7(5):541–546
Jacobson NS, Truax P (1991) Clinical significance: a statistical approach to defining meaningful change in psychotherapy research. J Consult Clin Psychol 59(1):12–19
Wyrwich KW, Tierney WM, Wolinsky FD (1999) Further evidence supporting an SEM-based criterion for identifying meaningful intra-individual changes in health-related quality of life. J Clin Epidemiol 52(9):861–873
Yost KJ, Cella D, Chawla A, Holmgren E, Eton DT, Ayanian JZ et al (2005) Minimally important differences were estimated for the Functional Assessment of Cancer Therapy-Colorectal [FACT-C] instrument using a combination of distribution- and anchor-based approaches. J Clin Epidemiol 58(12):1241–1251
Juniper EF, Guyatt GH, Willan A, Griffith LE (1994) Determining a minimal important change in a disease-specific Quality of Life Questionnaire. J Clin Epidemiol 47(1):81–87
Jaeschke R, Singer J, Guyatt GH (1989) Measurement of health status. Ascertaining the minimal clinically important difference. Control Clin Trials 10(4):407–415
Cella D, Eton DT, Lai JS, Peterman AH, Merkel DE (2002) Combining anchor and distribution-based methods to derive minimal clinically important differences on the Functional Assessment of Cancer Therapy [FACT] anemia and fatigue scales. J Pain Symptom Manag 24(6):547–561
Cohen J (1988) Statistical power analysis for the behavioral sciences, 2nd edn. Lawrence Erlbaum, Hillsdale
King MT (2011) A point of minimal important difference [MID]: a critique of terminology and methods. Expert Rev Pharmacoecon Outcomes Res 11(2):171–184
Conflict of interest
None declared. The authors declare that they have full control of all primary data. We agree to allow the journal to review the data if requested.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Den Oudsten, B.L., Zijlstra, W.P. & De Vries, J. The minimal clinical important difference in the World Health Organization Quality of Life instrument—100. Support Care Cancer 21, 1295–1301 (2013). https://doi.org/10.1007/s00520-012-1664-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-012-1664-8